If accepted, the breakthrough product will convey Cerapedics’ P-15 Peptide to lumbar spine fusion with a TLIF indication, making it Cerapedics’ second Class III drug/machine combo product
WESTMINSTER, Colo., Jan. 4, 2024 /PRNewswire/ — Cerapedics Inc., a commercial-stage orthopedics firm devoted to redefining the usual of look after bone restore, at the moment introduced that it has filed the ultimate module within the Pre-Market Approval (PMA) utility with the U.S. Food and Drug Administration (FDA) for its investigational P-15 Peptide Enhanced Bone Graft. Cerapedics was capable of advance to this submission sooner than anticipated resulting from using a rigorous statistical evaluation generally known as the Bayesian A number of Imputation. If accepted, this would be the newest bone graft product in Cerapedics’ portfolio to leverage its highly effective, proprietary P-15 Osteogenic Cell Binding Peptide, which has a confirmed mechanism of motion not present in some other class of bone graft supplies.
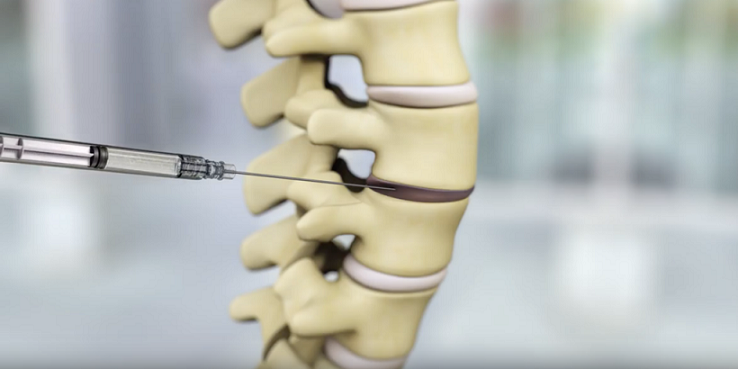
The brand new P-15 Bone Graft is designed to be used in transforaminal lumbar interbody fusion (TLIF) surgical procedure in sufferers with degenerative disc illness. In April 2021, the FDA granted breakthrough machine designation for the brand new P-15 Bone Graft supposed to be used within the therapy of a single-level TLIF in sufferers with degenerative disc illness. The designation provides a chance to work together with FDA consultants to deal with regulatory subjects effectively and permits for prioritized evaluation of regulatory submissions.
“We’re proud to submit our PMA utility, and to be nearer to introducing our revolutionary P-15 Peptide Enhanced Bone Graft to the big inhabitants of sufferers present process TLIF procedures within the U.S.,” stated Valeska Schroeder, Chief Govt Officer of Cerapedics. “With substantial Stage 1 human medical knowledge supporting the product, we stay up for our continued collaboration with the FDA within the upcoming evaluation course of.”
The PMA utility relies on knowledge from the ASPIRE research, a potential, single-blinded, multi-center, randomized, non-inferiority pivotal IDE research to judge the security and efficacy of P-15 Bone Graft in contrast to make use of of an autologous bone graft when utilized in TLIF surgical procedure. The ASPIRE trial included 33 US facilities and 290 sufferers, lots of which had been thought of excessive threat for non-union (diabetics, overweight sufferers, and nicotine customers). The first endpoint of the research checked out a 24-month composite medical success outlined as assembly fusion (assessed by way of skinny lower CT at 6, 12 and 24-months), operate (ODI), neurological, index degree secondary surgical procedure intervention, and critical device-related antagonistic occasion endpoints. Based mostly on assembly the necessities of a Bayesian evaluation, Cerapedics submitted the ultimate PMA module earlier than all sufferers reached the month 24 follow-up go to.
CAUTION: The brand new P-15 Bone Graft is an investigational product, restricted by United States legislation to investigational use.
About P-15 Osteogenic Cell Binding Peptide
Cerapedics revolutionary P-15 Peptide offers a mechanism of motion to Connect, Activate and Speed up exact* bone formation. P-15 Peptide is a 15 amino acid sequence discovered naturally in Kind 1 collagen. Cerapedics’ pharmaceutically manufactured model of P-15 Peptide is certain onto an osteoconductive scaffold offering an abundance of attachment websites for osteogenic, bone-forming, cells. P-15 Peptide reveals a excessive affinity for mobile attachment; as soon as osteogenic cells connect, intracellular processes that construct bone are activated, amplifying pure signaling, and accelerating new bone formation.
*Floor-bound mechanism of motion
About Cerapedics
Cerapedics is a worldwide, commercial-stage orthopedics firm that aspires to redefine the usual of look after bone restore by therapeutic bones quicker and at greater charges, with out compromising security, in order that sufferers can reside their healthiest lives. Bone grafts, together with Cerapedics’ merchandise, are utilized in over 4 million annual spine, orthopedics, trauma, and interventional procedures worldwide. i-FACTOR is a Class III FDA-approved product indicated for single-level ACDF. Cerapedics’ new “P-15 Bone Graft” product is at present being evaluated to be used in lumbar interbody fusion by way of ASPIRE, a pivotal medical research, and has been granted a Breakthrough Machine Designation by the FDA. Cerapedics is headquartered in Westminster, CO.
For extra data, go to us at www.cerapedics.com.
Media contact: Ten Bridge Communications
TBCCerapedics@tenbridgecommunications.com
SOURCE Cerapedics Inc.






















Discussion about this post